Scientific
newsletters

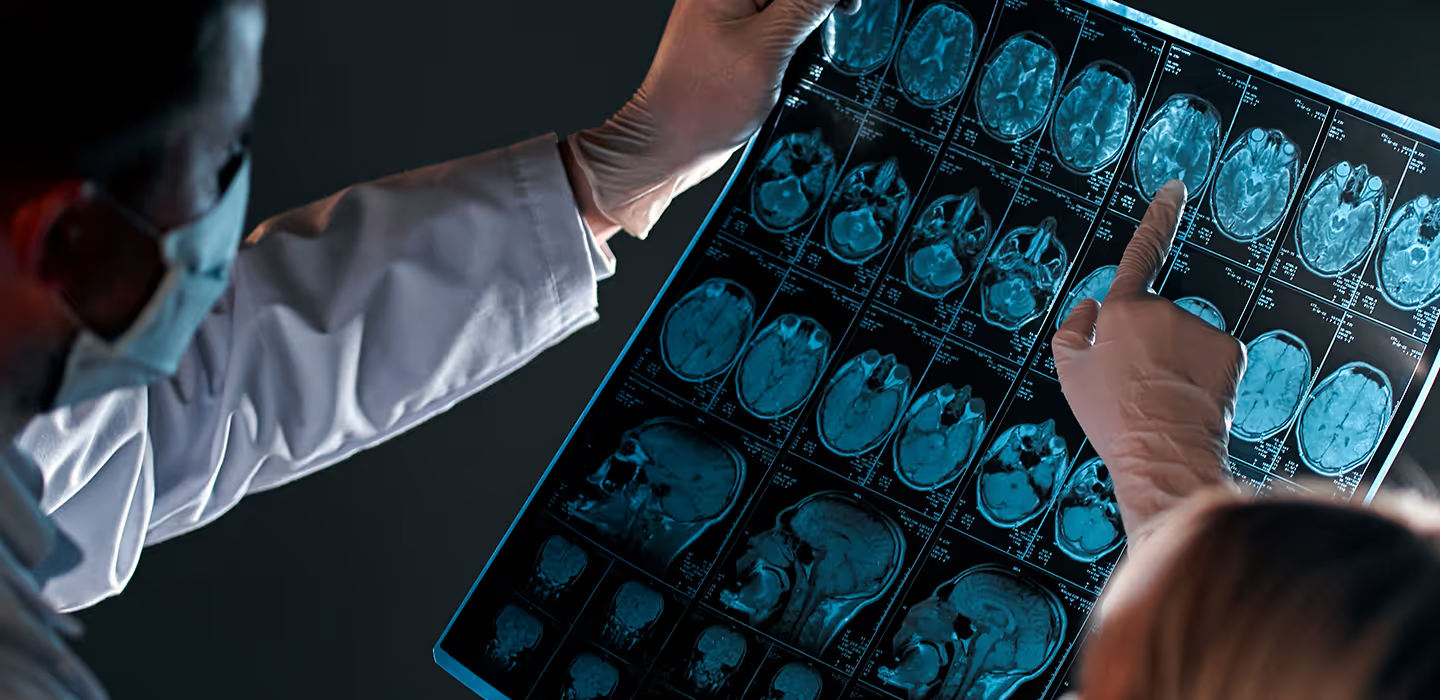
Magnetic resonance imaging of the brain in multiple sclerosis (MS) was presented for the first time on 14 November 1981, when “The Lancet” published an article entitled “Nuclear Magnetic resonance imaging of the brain in multiple sclerosis”, by Y. R. Young and his collaborators. These authors showed that CT (cranial X-ray computed tomography) scans of 10 patients with MS showed a total of 19 lesions altogether, while a further 112 lesions were shown on the MRI scans!
This was the first time, in other words, that neurologists were able to ascertain the spread of the disease and the number of brain lesions during a patient’s lifetime (the patient’s “lesion load”). The team soon realised that most of these lesions were asymptomatic, in ‘silent’ areas of the brain.
Significant improvements have since been made in terms of the quality of the images and their resolution, by increasing the field strength of the magnets used (0.5 then 1.5 and finally 3 Tesla) and adapting this technique to the spinal cord. This gave us the opportunity to distinguish between old lesions and active inflammatory lesions (new lesions or reactivated old lesions) associated with blood-brain barrier leakage. A paramagnetic contrast product called gadolinium leaks into the active lesions. An active lesion may eventually (but not always) result in a permanent central necrosis called “black hole”, due to demyelination and transsections of the nerve fibres.
This made it possible to distinguish between old lesions from active inflammatory lesions.
Since 2001, MRI has been used as part of the diagnostic criteria, based on the number and location of lesions, whether periventricular, cortical or juxtacortical, or in the cerebellum, brainstem or spinal cord. Finally, it also demonstrated something that neuropathology had already taught us, namely the presence of a central vein within MS lesions. The subpial demyelination sites, directly underneath the meninges, are still difficult to detect with conventional MRI.
Based on these images, prognostic criteria were formulated. Patients with lesions in the brainstem, cerebellum or lateral columns of the spinal cord have a poorer prognosis thanpatients who only have periventricular and hemispheric lesions. Some (but not all) lesions have been detected within the cerebral cortex, in addition to those located in the white matter (myelinated fibres). Disease-induced brain atrophy can now be measured beyond the normal mean brain volume loss below 0.4% per year. More selective and localised atrophies also occur, in the corpus callosum for example, a bundle of myelinated nerve fibres that connects the two brain hemispheres, the thalamus and the cervical spinal cord. More recently, MRI revealed focal areas of meningitis corresponding with the presence of ectopic lymphocytic nodules in the meninges.
Slowing down this brain atrophy to the normal values observed in every human being is the goal pursued in several recent studies of new treatments
Thanks to MRI, scientists were also able to demonstrate the partial, albeit significant, effectiveness of Betaferon, the first interferon used to treat this disease. It prevented the development of new lesions, in addition to reducing the clinical relapse rate. This first study, which was published in 1993, did not yet use the contrast product gadolinium, which became the gold standard in all subsequent studies. Global brain atrophy, which is another reliable outcome measure of drug effectiveness, was first used in trials of Fingolimod (Gilenya). Slowing the speed of brain atrophy to the normal levels that are observed in any person was the desired (and in some instances achieved) goal in several recent trials of new treatments.
To date, conventional MRI is not very quantitative in terms of the overall lesion volume and the presence of active chronic lesions that progress insidiously without breaking down the blood-brain barrier. The latter, however, are very important in progressive MS. More recently, studies demonstrated that they are partially or totally surrounded by a thin border of iron-containing inflammatory cells.
As Dr Solène Dauby explains, however, this technology can bring us new data that will increase our knowledge of this disease, for example by using MRI systems with a magnetic field of 7 Tesla. This will make it possible to analyse and quantify loss of nerve cells, the rarefaction of synaptic connections, decreased nerve fibre density and, potentially, the remyelination of some of them, whether spontaneously or thanks to new treatments that are currently being tested in clinical trials.
Prof. Dr Christian Sindic

Newsletter 50
Stay informed
Receive all the information related to research and news from the Belgian Charcot Foundation directly in your inbox.